Oncoproteins of Primate Rhadinoviruses
Proliferation, differentiation, and effector functions of T lymphocytes are regulated by an intricate network of multiple signaling pathways. They are triggered by ligation of the antigen-specific T-cell receptor (TCR), costimulatory receptors, cytokines, and chemokines. Dysregulated signaling is observed in diseases ranging from immunodeficiency to autoimmunity and T-lymphoid malignancies. We study T‑lymphotropic rhadinoviruses, Herpesvirus saimiri (HVS) and Herpesvirus ateles (HVA), and their oncoproteins that support transformation of primary human T cells to permanent growth in culture. Our analyses aim at the signaling pathways influenced by the viral oncoproteins to understand their role in regulation of T-cell proliferation and viral persistence.
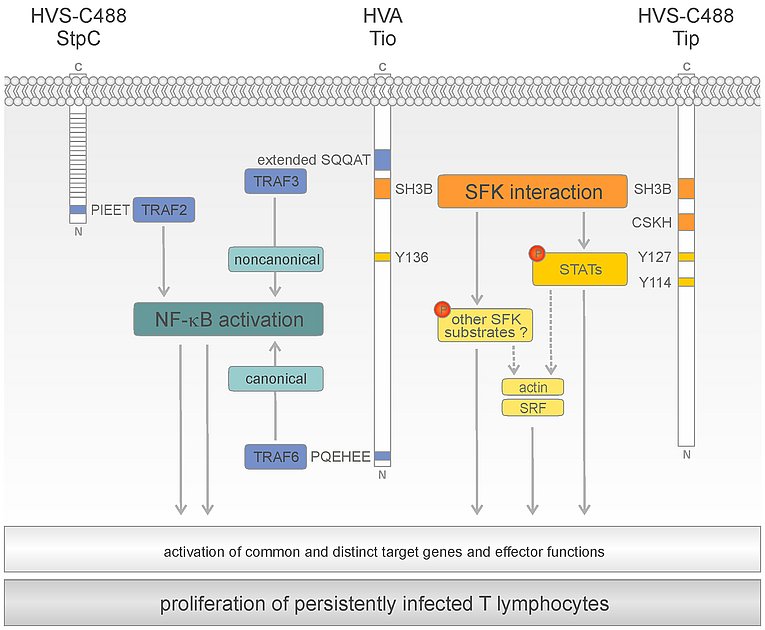
The HVS oncoproteins StpC and Tip as well as the HVA oncoprotein Tio bind to Src-family kinases (SFK) and NF-κB-regulating TRAFs through distinct amino acid motifs. Accordingly, both SFK and NF-κB activity is required to maintain the growth-transformed phenotype of persistently infected T-cells.
Interactions with Src-family kinases (SFK)
The Tyrosine kinase interacting protein (Tip) of HVS strain C488 was identified as a phosphoprotein associated with Lck, a major T-cell specific SFK. Binding of Tip to Lck is based on modular interactions with the SH3, kinase, and SH2 domains. Similar to Tip, the Two-in-one (Tio) oncoprotein of HVA interacts with the SH3 and SH2 domains of several SFKs, including Lck and Lyn. Integrity of the SFK binding motifs in Tip and Tio is crucial for viral T-cell transformation. Specifically, targeting of SFK SH3 domains correlates with the viral capacity to transform human T lymphocytes.
Dependent on their SFK interaction sites, Tip and Tio induce the activation-specific phosphorylation of STAT1, STAT3, and STAT5. In addition, activation of STAT1 and STAT3 by Tip relies on tyrosine residue 114 (Y114), which may serve as an Lck substrate and, subsequently, as a STAT binding site. However, Tip mutant Y114F still supports viral T-cell transformation in culture. Likewise, activation of serum response factor (SRF) by Tip depends on SFK interaction as well as Y114. Thus, signaling intermediates, which are downstream of SFK SH3 binding and essential for viral transformation, remain to be identified.
Interactions with TRAFs and NF-κB activation
The Saimiri transformation-associated protein of subgroup C (StpC) harbors an N‑terminal binding site for TNF receptor-associated factors (TRAFs), especially TRAF2, that is required for both canonical and noncanonical NF‑κB activation. In contrast, Tio induces either of the two NF‑κB pathways through a distinct molecular mechanism. Recruitment of TRAF6 to the N-terminal PQEHEE motif efficiently triggers canonical NF-κB activity. Inhibition of this pathway results in death of virus-transformed T lymphocytes. Recruitment of TRAF3 to a unique, extended TRAF binding motif is highly selective and induces noncanonical NF‑κB activity without affecting the canonical pathway. Therefore, Tio represents a unique tool to study noncanonical NF-κB signaling in T-cells.
Induction of MMP9 by Tio
In an attempt to identify cellular genes induced by Tio in dependance of noncanonical NF-κB signaling, we identified MMP9 as a potential candidate. Ongoing analyses indicate that Tio relies on the activity of canonical and noncanonical NF-κB as well as SFKs to upregulate this matrix metalloprotease involved in multiple biological processes including tumor cell invasion.
Induction of IFN-γ
Human lymphocytes transformed by H. saimiri subgroup C closely resemble mature, activated T-cells with a cytokine expression pattern skewed towards a Th1 phenotype. Accordingly, we identified the typical Th1 cytokine, interferon γ (IFN-γ), as a downstream target of the viral oncoproteins. While Tip-SFK interaction is essential for the basal induction, StpC augments IFN-γ expression through NF-κB activation. Recent experiments demonstrate that Tio also triggers IFN-γ production through SFK as well as canonical and noncanonical NF-κB activity. However, STAT1 phosphorylation depends on SFK interaction only and, thereby, excludes autocrine signaling. Thus, the role of IFN-γ in viral T-cell transformation in culture remains enigmatic. In infected animals, however, virus-induced IFN-γ production might contribute to viral pathogenesis.