Gene-based immunization strategies
Without any doubt, vaccines are the most effective measures to prevent infectious diseases and to promote individual and public health. Our group has developed and optimized gene-based vaccines for viral respiratory pathogens, including SARS-CoV-2, Respiratory Syncytial Virus (RSV) and Influenza A Virus (IAV). During the recent SARS-CoV-2 pandemic, the enormous potential of mRNA and adenoviral vector vaccines to induce broadly protective immunity was demonstrated for the first time in humans. The licensed COVID19 vaccines are applied intramuscularly and provide excellent protection against severe disease, hospitalization and death, but do not effectively prevent breakthrough infection in and transmission among vaccinated individuals. These findings are consistent with our previous knowledge from viral respiratory tract infections in animal models, which guided our research interest to the development of vaccines able to induce immunity at the viral entry site, the mucosa of the respiratory tract. Here are some highlights of our recent research activities:
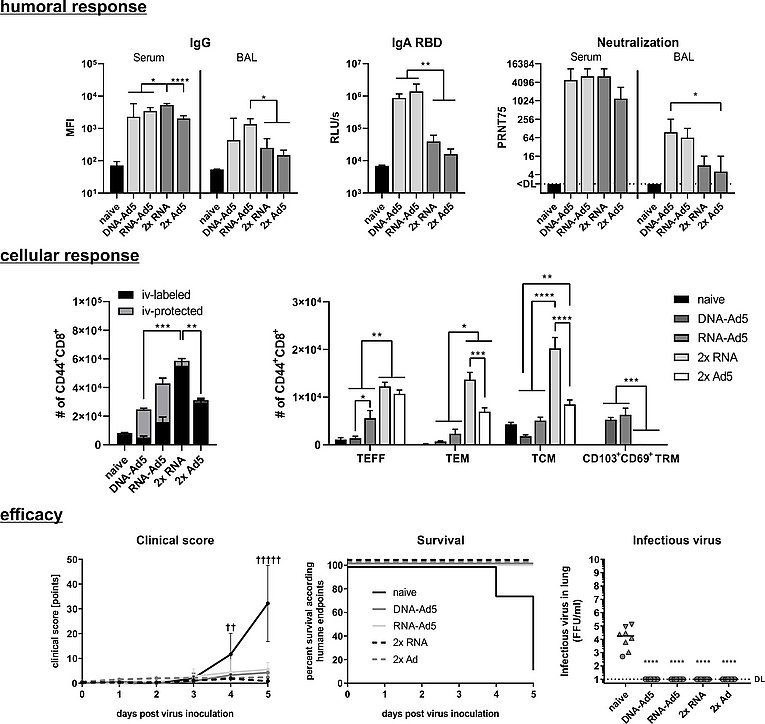
C57BL/6 mice received an intramuscular prime immunization with the spike-encoding DNA, Ad5-S, or the mRNA vaccine, Comirnaty®. Mice from the heterologous prime-boost groups were boosted four weeks later intranasally with Ad5-S. The homologous prime-boost groups received a second dose of mRNA or Ad5-S intramuscularly.
Upper panel: Spike-specific IgG antibodies in serum and BAL; RBD-specific IgA in BAL and neutralizing antibodies in serum and BAL
Middle panel: Antigen-experienced CD8+ memory T-cells were identified by CD44 staining and differentiate between circulating (iv-labelled) and tissue-resident (iv-protected). Further, effector T-cells (TEFF), effector memory T-cells (TEM), and central memory T-cells (TCM) were defined within the circulating T-cells, whereas TRM cells were defined as KLRG1-CD103+CD69+.
Lower Panel: K18-hACE2 mice were challenged four weeks after the immunization with with 9x103 FFU SARS-CoV-2. All animals were monitored daily for clinical score and survival. Animals reaching humane endpoints were euthanized and are marked by a cross (†).Infectious virus was retrieved and titrated from lung homogenates.
Mucosal immunity against viral respiratory tract infections induced by gene-based immunizations
Along the newly emerged SARS-CoV-2, established pathogens such as IAV and RSV are causative agents of severe respiratory tract infection especially in young children and elderly people. The global disease burden is estimated to ~ 650 million cases per year for these two viruses leading to estimated 0.5 million deaths/year worldwide. Due to the short incubation period of respiratory viral infections, vaccines need to induce immune responses yielding an immediate antiviral response upon exposure. Secretory IgA and tissue-resident memory T-cells (TRM) at mucosal surfaces have been identified as major players involved in the protection against pulmonary virus infections. Specifically, lung TRM cells directed against the conserved nucleoprotein of IAV provide heterosubtypic immunity against divergent IAV strains. Since TRM cells reside in the target tissue, they locally respond to a secondary infection, restrict immediate viral replication and recruit further immune cells by the secretion of inflammatory cytokines, e.g. IFN-γ.
Fitting to the observation that intramuscularly applied vaccines do not induce mucosal T-cells, our research confirmed local antigen presentation and inflammation induced by mucosal vaccines as prerequisite for the induction and imprinting of lung TRM (Lapuente et al., 2018b). We evaluated different vaccine platforms, including DNA, RNA or replication-deficient adenoviral vectors (Ad5/Ad19a) for their potential to induce local antibody and T-cell responses and to provide protection from IAV, RSV and SARS-CoV-2 in mice (Lapuente et al, 2018a; Lapuente el al., 2021b).
However, these studies also confirmed that a prior systemic immunization with either DNA or RNA based vaccines encoding the same viral antigens maximizes the immunogenicity of the mucosally applied adenoviral vector vaccines. An intramuscular DNA immunization followed by an mucosal adenoviral boost resulted in mucosal immunity, including antiviral IgA antibodies in the BAL and TRM responses, and provided efficient protection against Influenza and RSV in mice and NHP, respectively (Lapuente et al, 2018a; Grunwald et al., 2014). A confirmatory, multi-center non-human primate study as a next step to translate this approach into a clinical trial is currently on-going.
Most recently, we utilized the licensed mRNA vaccine Comirnaty® as systemic prime immunization followed by intranasal vaccinations with adenovirus 5 and 19a vectored vaccines encoding the SARS-CoV-2 spike protein (Lapuente el al., 2021b). In contrast to two intramuscular applications of the mRNA vaccine, intranasal boosts with adenoviral vectors induced high levels of mucosal IgA and lung-TRM. The mucosal neutralization of virus variants of concern was also enhanced.
The mRNA prime provokes a comprehensive T-cell response consisting of circulating and lung TRM after the boost, while the repeated intramuscular immunization induced almost exclusively circulating, non-resident memory T-cells. Concomitantly, the intranasal boost strategies lead to complete protection against a SARS-CoV-2 infection in mice (Fig.1).
Having established this efficient combination of a systemic prime-mucosal boost vaccination schedule, we are now interested in how we can further optimize the mucosal booster to induce broad and long-lived mucosal immunity. This project is designed and will be undertaken as part of a Bavarian research consortium named FOR-COVID.
Signal molecules of the innate immune system as genetic adjuvants
One way to improve the immunogenicity of vaccines is the use of adjuvants. To allow temporal and spatial co-delivery of the adjuvants with the antigen, we investigated adenoviral vectors encoding various chemokines or cytokines as potential adjuvants for the induction of mucosal immunity.
So far, the co-delivery of an adenoviral vector encoding IL-1ß dramatically increased the numbers of IAV-NP-specific CD103+ CD69+ TRM in the lung, which translated in superior protection against divergent IAV strains, such as pH1N1, H3N2, and H7N7 (Lapuente et al., 2018b). In the context of RSV, the Ad-IL-1β adjuvanted, adenoviral vector vaccine conferred even superior protection against secondary RSV infection compared to natural immunity induced by a primary RSV-infection in mice (Maier et al, 2022). Most recently, in a collaborative study with the Pirbright Institute, we could confirm the adjuvant effect of Ad-IL-1β on the induction of cross-reactive TRM in the highly relevant large animal model of pigs. Interestingly, the increased number of porcine, NP-specific TRM cells had no significant impact on a heterologous IAV infection (Schmidt et al, 2023).
Next, we would like to decipher potential differences in the correlates of hetero subtypic immunity between different animal models.
Induction and fate of tissue-resident memory T-cells
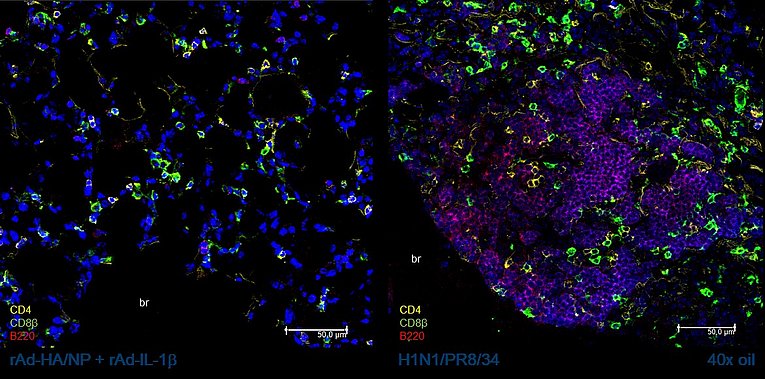
The importance of lung-resident memory T-cells for the rapid control of viral respiratory tract infections has been well documented. The programming of antigen-specific T-cells to TRM seems to happen early during the priming phase and depend very much on the local inflammatory environment and antigen presentation. In contrast, the mechanisms of the maintenance of TRM in this immune privileged tissue are not yet fully understood. We determine whether the differential induction of the TRM immunity by a primary infection or by the mucosal application of gene-based vaccines might play a role in this context. In longitudinal studies, phenotypic and functional TRM responses are assessed in mice. Furthermore, the structural organization within the tissue will be examined via immunohistology (Fig. 2) and the differential transcriptional programming will be analyzed by single cell RNA sequencing. Finally, we will particularly address the impact of secondary inflammatory events, such as unrelated bacterial or viral infections, on the fate of pre-existing, virus-specific TRM.
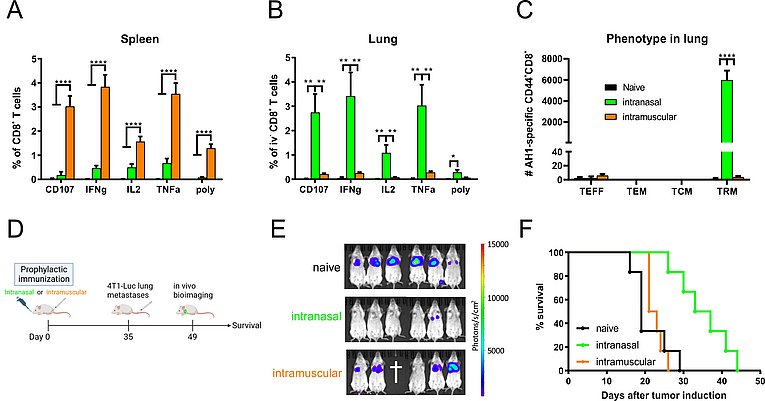
Mice were immunized intranasally or intramuscularly with the adenoviral 4T1 vaccine. Five weeks later, AH1-specific T-cell responses were measured in spleen (A) and lung (B) by peptide restimulation and intracellular cytokine staining. The memory phenotype of lung T-cells was characterized with AH1 multimers and staining of CD44, KLRG1, CD127, CD69 and CD103 (C). T-cell responses in (B) and (C) were pregated on cells negative for intravenous staining (iv-). Vaccine efficacy was evaluated in a prophylactic setting by in vivo bioimaging two weeks after 4T1-Luc tumor induction (E) and by continuous survival monitoring. p < 0.05 vs. naive; *p < 0.05; **p < 0.005; ***p < 0.0005; ****p < 0.0001
A mucosal vaccination strategy to prevent and treat lung-metastasized breast cancer
(PI: Dr. rer. nat. Dennis Lapuente)
Breast cancer is the most prevalent cancer worldwide with 2.3 million diagnoses each year accounting for 25% of all malignancies in women. In most cases, an early diagnosis yields a relatively good prognosis (5-year survival of 99%). The presence of metastases in distant organs including the lung worsens the prognosis significantly (5-year survival 29%). Our goal is to develop a mucosal vaccination strategy to inhibit the formation and growth of pulmonary breast cancer metastases.
We have designed an adenoviral vector vaccine in the preclinical 4T1 breast cancer model for the induction of CD8+ T-cell responses directed against two endogenous tumor-associated antigens (gp70 AH1, Mage-b). Depending on the route of vaccine administration, the vaccine establishes either robust systemic immunity after intramuscular or pronounced TRM responses after intranasal delivery (Fig. 3 A+B). Vaccine-induced TRM display a prototypical CD69+CD103+ phenotype (Fig. 3 C) and remain unstained upon in vivo intravenous antibody staining (a method to distinguish circulating from resident immune cells). In a prophylactic setting (Fig. 3 D), the mucosal vaccination inhibited the tumor growth more efficiently than a systemic immunization highlighting the importance of mucosal immunity at barrier sites (Fig. 3 E+F). Currently, we are characterizing this vaccine approach in a therapeutic setting. Moreover, we want to understand synergistic effects of therapeutic vaccination and clinically relevant treatment options such as immune checkpoint blockade or chemotherapy. Together with Prof. Udo Gaipl (Radiation Oncology, University Hospital Erlangen), we are also investigating a combination of radiotherapy and mucosal immunization.
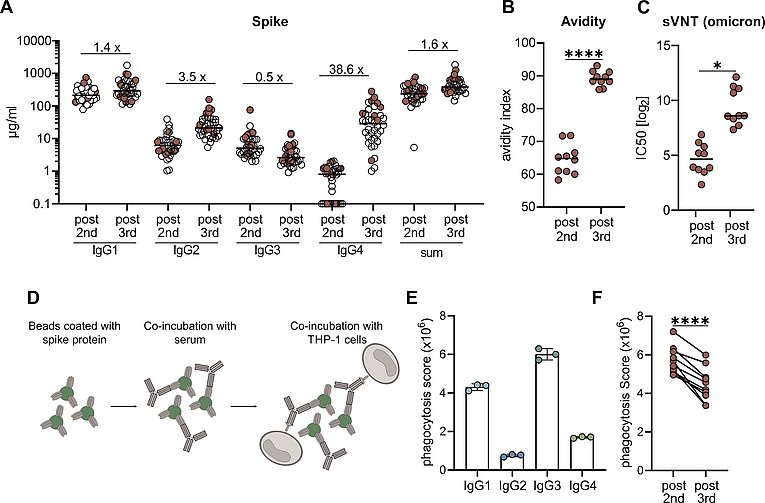
From a cohort of 38 vaccinees the IgG subclasses distribution was measured in a flow cytometric assay and the sum of all IgG are shown for the whole cohort (A). Paired serum samples from ten persons collected post 2nd or post 3rd vaccination were further analysed in regard to avidity (B) and neutrasing capacity against omicron (C).. Antibody-dependent phagocytosis by the monocytic THP-1 cell line was analysed by using either monoclonal RBD antibodies of the different subclasses or the paired sera (D-F).
Antibody response to SARS-CoV-2
In the early phase of the pandemic, we established serological assays to analyze antibody responses to SARS-CoV-2 after infections. A flow cytometry-based assay allows us to quantify antibodies of the different Ig isotypes (IgM, IgA, IgG) binding to SARS-CoV-2 spike (S) protein (Lapuente et al., 2021a). In several studies, we analyzed the SARS-CoV-2 specific immune responses for example in cohorts with different immunization schedules (Tenbusch et al, 2021; Vogel et al, 2022) or in specific cohorts of patients receiving immunomodulatory medical interventions (Simon et al, 2020; Reimann et al, 2022; Kremer et al, 2021).
Together with the groups of Prof. Thomas Winkler and PD Kilian Schober, we observed an atypical increase in vaccine-induced IgG4 antibody responses against the SARS-CoV-2 spike protein after three doses of the mRNA vaccine Comirnaty (Irrgang et al, 2023). The contribution of virus-specific antibodies in preventing infections and disease progression depends beside neutralizing activity very much on secondary effector functions mediated by binding to Fc-gamma receptors (FcgR) on various immune cells. In this regard, IgG4 represents an IgG isotype with low potential of mediating antiviral effector functions such as antibody-mediated cellular cytotoxicity (ADCC) or phagocytosis (ADCP) and is considered as anti-inflammatory (Fig.4).
In the context of viral infectious diseases not much is known about the contribution of this rare subclass. Our exciting discovery in the field of immunology raises new questions about antibody maturation and the consequence of strong IgG4 responses in preventing viral infection or disease progression. In our recent projects, we want to address these questions in appropriate small animal models.