Nanoparticle vaccines
Vaccination is the most effective method to achieve long-lasting protection against infectious diseases. Nanoparticle vaccines, a novel type of vaccine that can be composed of various materials like lipids, polymers, or inorganic molecules. Numerous studies indicate their potential to enhance immune responses against a wide range of infectious diseases. The intrinsic properties of nanoparticles, in synergy with advancements in genetic and biochemical modification techniques, empower nanoparticle-based vaccines as an innovative and transformative approach to rational vaccine design. The Nanoparticle vaccines group’s research goal is to contribute to the development of innovative vaccination strategies by designing, characterizing, and conducting preclinical evaluations of diverse B-cell targeting nanoparticle-based prototype vaccines. We are currently working on the following main types of nanoparticles:
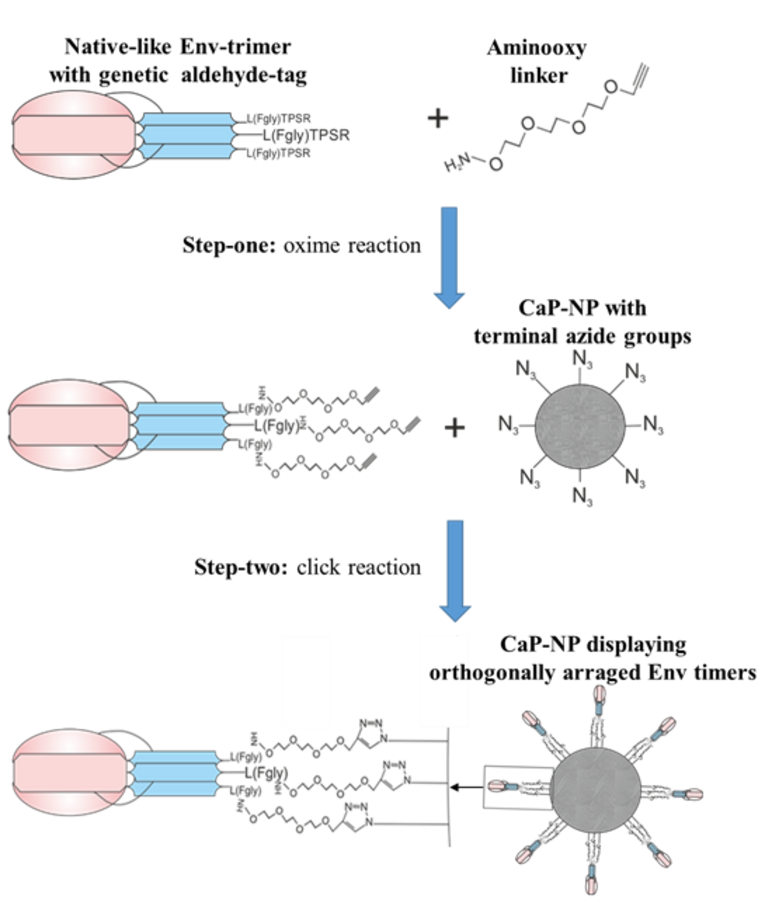
Step-one: Env trimers with aldehyde-tags at the C-termini were bound to linker molecules with both aminooxy- and alkyne-terminated reactive groups and a small PEG spacer in-between. Step-two: the alkyne groups of the resulting linker-bound Env can be harnessed for copper-catalyzed azide-alkyne cycloaddition („click reaction“) to CaP nanoparticles with terminal azide groups.
Biodegradable inorganic calcium phosphate nanoparticles
During the last decade, antigen-functionalized nanoparticles have become a major focus in the field of HIV-1 vaccine research. The efficacy of an HIV-1 vaccine, however, depends on the presence of vaccine-induced immune mechanisms that may increase the susceptibility for HIV-1 infection. The unique characteristic of HIV-1 to infect activated CD4+ T-cells requires alternative vaccination strategies that differ from the classical approaches used. In particular, the number of HIV-specific IFN-γ-secreting cells induced by vaccination correlates with an increased risk of infection. Currently, in cooperation with Prof. Matthias Epple we designed biodegradable calcium phosphate nanoparticles (CaP-NP) with stabilized HIV-1 envelope trimers coupled covalently in a randomly oriented manner to the surface of CaP-NP (rEnv-CaP-NP). We demonstrated improved B-cell activation in the presence of nanoparticle-bound antigens compared to soluble trimers in vitro. To avoid the induction of HIV-1 specific CD4+ T helper (Th) cells we recruited heterologous T cell responses previously induced by a licensed vaccine via the intrastructural help (ISH) mechanism. To this end, we encapsulated universal helper peptides of tetanus toxoid in the core of rEnv-CaP-NP. This incorporation of T helper epitopes derived from heterologous (non-HIV) proteins elevated the magnitude of anti-Env specific IgG antibody responses by ISH without significant induction of Env-specific IFN-γ-secreting Th cells in a small animal model. This suggests that the ISH strategy for calcium phosphate nanoparticle-based HIV-1 vaccines may allow bypassing the induction of HIV-1-specific IFN-γ-secreting Th cells suspected to enhance the susceptibility for HIV-1 infection.
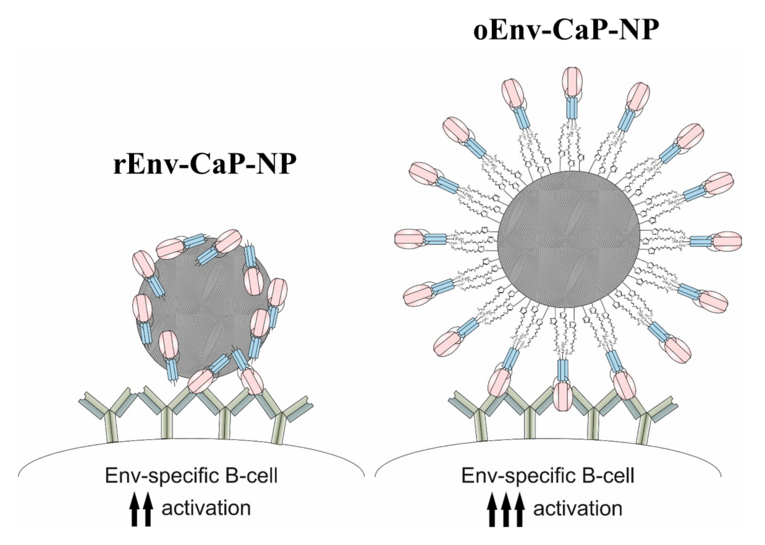
By the random coupling of Env trimers, however, the exposition of the non-glycosylated base of these soluble antigens may bias immune responses, since this immunodominant region is an easy target for non-neutralizing antibodies. An orthogonal array of Env trimers on a nanoparticle carrier could occlude the base of the trimer and enhance B cell activation, but the coupling procedure must guarantee to maintain the protein conformation. Various molecular mechanisms to couple native-like trimers of the HIV-1 envelope protein onto nanoparticle surfaces have been reported, but many come with disadvantages regarding the coupling efficiency and stability. We used a recombinant aldehyde-tag for the site-specific coupling of conformational Env trimers onto the surface of nanoparticles.
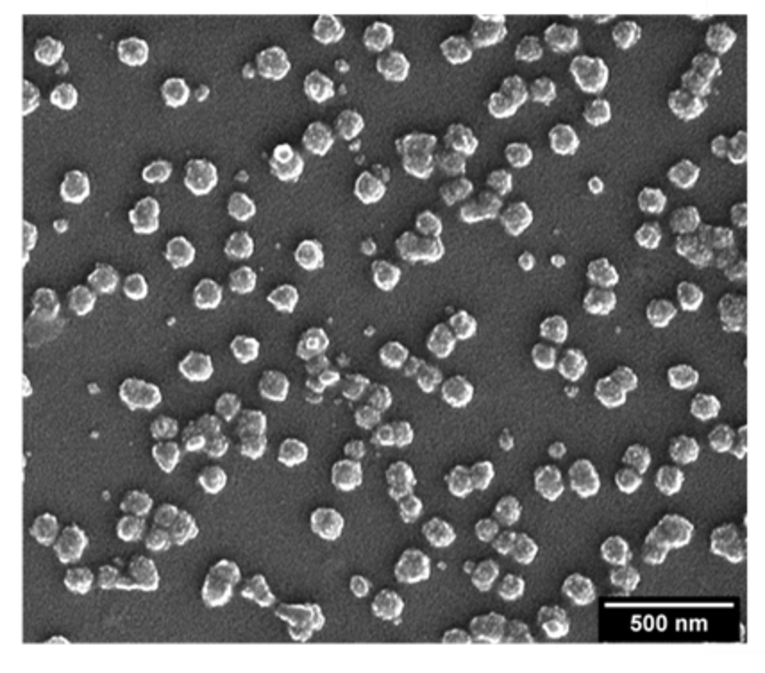
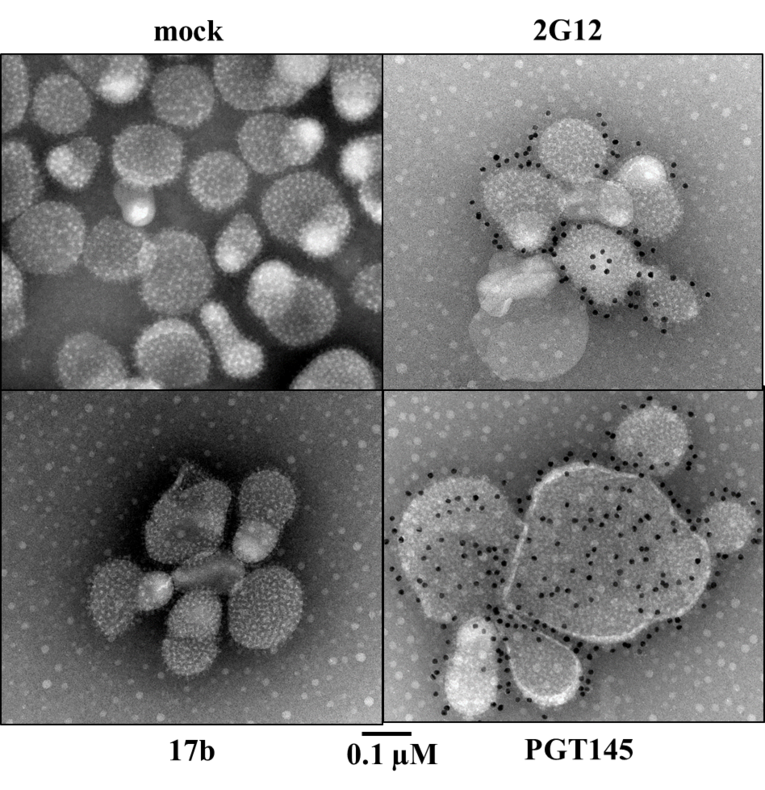
In follow-up experiments, we were able to demonstrate the efficiency of oxime ligations and click reactions. The preservation of the trimer conformation on the surface of CaP-NPs after orthogonal coupling (oEnv-CaP-NP) was clearly demonstrated with an experimental flow cytometry approach using conformation-specific monoclonal antibodies. Scanning electron microscopy analysis showed a spherical morphology of oEnv-CaP-NP.
oEnv-CaP-NP display more accessible, identical antigen epitopes on their surface in comparison to rEnv-CaP-NP. This may facilitate a better recognition by BCRs of Env-specific B cells and improved BCR crosslinking under conditions of limited antigen amount.
oEnv-CaP-NPs displaying orthogonally oriented Env trimers on their surface were superior in activation of Env-specific B-cells (in vitro) and induction of anti-Env specific antibodies (in vivo) compared to rEnv-CaP-NPs with Env trimers coupled in a randomly oriented manner.
Thus, covalent orthogonal coupling of Env trimers offers important foundations for the rational design of nanoparticle-based HIV-1 vaccine platforms. The high efficiency of aldehyde-mediated Env coupling without the need for toxic catalytic reagents renders the strategy feasible for clinical applications. The presented approach may be generalized to other viral glycoproteins displaying quaternary structure epitopes such as pre-fusion stabilized spike proteins of coronaviruses, stabilized trimeric glycoproteins of Ebola and Marburg filoviruses, and stabilized E-protein dimers of Zika and Dengue flaviviruses with applications in diagnostics and vaccine development.
T helper liposomes
Current advances in conjugating native-like HIV-1 envelope trimers onto liposomes and encapsulating peptide epitopes into these nanoparticles have rendered this GMP-scalable vaccine platform a competitive alternative to other nanoparticle-based vaccines. In cooperation with Prof. Karola Vorauer-Uhl we designed and functionally characterized customizable Env-conjugated T helper liposomes (lipid nanoparticles that display the B cell-targeting Env antigen on the surface and contain T helper cell epitopes in the core). We passively encapsulated T helper peptides into a well-characterized liposome formulation displaying a dense array of Env trimers on the surface. We confirmed the closed pre-fusion state of the coupled Env trimers by immunogold staining with conformation-specific antibodies.
These peptide-loaded Env-liposome conjugates efficiently activated Env-specific B cells, which further induced proliferation of CD4+ T cells by presentation of liposome-derived peptides on MHC-II molecules. The peptide encapsulation process was then quantitatively improved by an electrostatically driven approach using an overall anionic lipid formulation. We demonstrated that peptides delivered by liposomes were presented by DCs in secondary lymphoid organs after intramuscular immunization of mice. UFO (uncleaved prefusion optimized) Env trimers were covalently coupled to peptide-loaded anionic liposomes by His-tag/NTA(Ni) interactions and EDC/Sulfo-NHS crosslinking. The conformation of the trimers was verified by liposomal surface flow cytometry. Furthermore, anionic Env-coupled T helper liposomes effectively induced Env-specific B cell activation and proliferation.
We further functionalized cationic liposomes with Env trimers by a tag-free approach designed to enable a simplified GMP production. The pre-fusion conformation of the conjugated Env trimers was verified by electron microscopy imaging and flow cytometry. The cationic liposomes induced strong activation of Env-specific B cells in vitro. In comparison to previously described anionic liposomes, cationic T helper liposomes were superior in CD4+ T cell activation after uptake by dendritic cells. Moreover, the T helper liposomes were able to target Env-specific B cells in secondary lymphoid organs after intramuscular injection. We also observed efficient T helper cell activation and proliferation in co-cultures with Env-specific B cells in the presence of cationic T helper liposomes.
Taken together, we generated T helper liposomes by different approaches. An array of in vitro methods was established and adapted to functionally characterize these nanoparticles in terms of Env trimer conformation, quantitative peptide encapsulation, activation of Env-specific B cells as well as the uptake and peptide presentation to T helper cells. The established functional nanoparticle characterization assays can be applied to other systems of synthetic nanoparticles delivering antigens derived from various pathogens.
HIV-derived virus-like nanoparticles
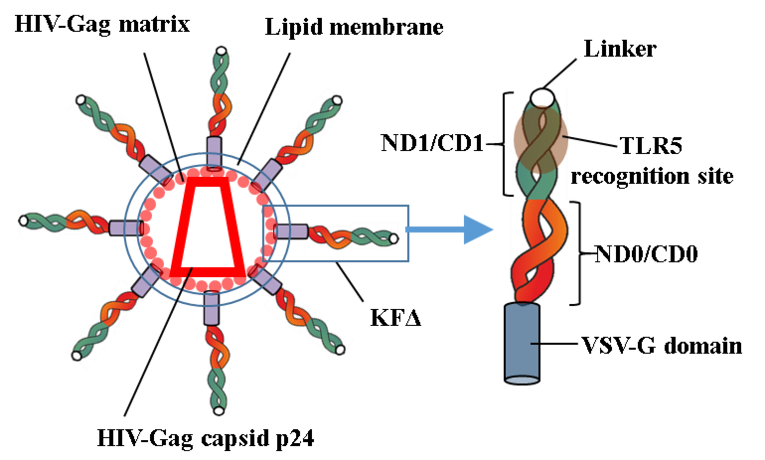
Virus-like particles (VLPs) are a promising tool for HIV-1 vaccine development.The great advantage of VLP-based vaccines is their structural identity to wild-type viruses, ensuring that antigen-specific B-cells encounter viral proteins in their natural conformation. VLPs can be further genetically or biochemically functionalized with biomolecules (antigens and adjuvants). HIV-derived enveloped VLPs consist of a common HIV-Gag protein backbone enclosed in a cellular lipid membrane in which the proteins of interest can be expressed. Previously, we functionalized HIV-derived VLPs with model antigens that were originally soluble, non-viral proteins. We used a genetic approach to integrate a truncated form of bacterial flagellin of a nonpathogenic E. coli strain that lacks the D2 and D3 domains into HIV-based viral nanoparticles via recombinant fusion with the transmembrane domain of the vesicular stomatitis virus G protein (VSV-G). Deletion of hypervariable domains ND2–D3–CD3 reduced the immunogenicity of the protein and the systemic inflammatory response against it, but retained the TLR5 agonist activity. An increasing number of studies have demonstrated the efficiency of flagellin as an adjuvant, as well as its ability to promote cytokine production by a range of innate immune cell types. Moreover, it triggers the recruitment of T- and B-cells to secondary lymphoid sites and activates dendritic cells (DCs) and T lymphocytes. Previously, using CaP-NP B-cell-targeting vaccines functionalized with a model antigen, hen egg lysozyme (HEL), we demonstrated that additional functionalization with flagellin improved antibody responses against HEL in mice. Here, we generated HIV-based VLPs displaying multiple molecules of the membrane-bound form of truncated flagellin (KFΔ) with an orientation optimized for TLR5 recognition.
The rational design of the KFΔ molecule prevents precipitation of nanoparticles and exposes the TLR-5 binding site outwards. KFΔ-functionalized HIV-based VLPs demonstrated bioactivity in vitro. Functionalization of B-cell-targeting VLP vaccines with KFΔ demonstrated both inhibitory and adjuvantive immunostimulatory effects on humoral immune responses against target antigens. The overall outcome of immunizations was based on the immunogenicity balance between the antigen and KFΔ. Therefore, in contrast to other TLR-ligands, the antigenic property of flagellin in comparison to the immunogenicity of the target antigen has to be considered for the functionalization of viral nanoparticle vaccines.