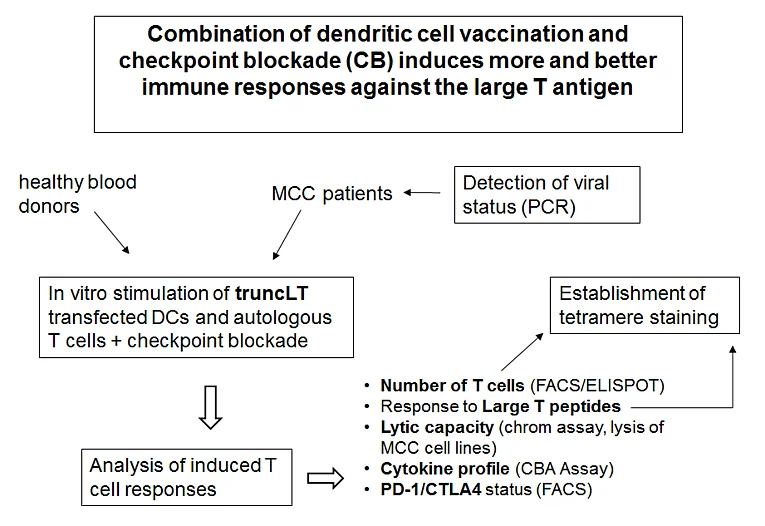
Preclinical evaluation of a combination of checkpoint blockade and dendritic cell vaccination against Merkel Cell Carcinoma
Merkel Cell Carcinoma (MCC) is a rare and highly aggressive skin tumor caused by the usually non-oncogenic Merkel Cell Polyomavirus. Under certain circumstances this virus integrates into the host cells` genome and expresses a truncated form of one of its proteins, the large T antigen (truncLT). Apart from surgical excision as well as radio- and chemotherapy, the treatment with checkpoint inhibitors such as anti-PD-1/PD-L1 (programmed death 1 receptor/PD-ligand 1) and anti-CTLA-4 (cytotoxic T-lymphocyte-associated Protein 4) has shown promising results in metastatic MCC. These antagonistic antibodies target inhibitory receptors on T cells and their ligands to enhance anti-tumor immune responses. Nevertheless, not all patients respond to this checkpoint blockade, so that additional treatment options have to be discovered. One possible option to make the tumor visible for the immune system is the principle of dendritic cell-based therapeutic vaccination which is already evaluated in several clinical trials. In general, dendritic cells (DCs) process and present antigens (Ags) and control Ag-specific T cells and their differentiation. Therefore, ex vivo generated DCs loaded with viral target antigens could be used to treat virally induced cancers.
The aim of this project is to evaluate a possible combination of checkpoint inhibitors and therapeutic vaccination against MCC and other virally induced cancers. Our hypothesis declares that this combination induces better immune responses against the truncLT than each treatment alone. To show this, we will analyze the influence of the different checkpoint-blockade antibodies alone and in combination in human ex vivo cell culture systems, in which the immunogenicity of DCs towards autologous T cells is elucidated (figure 1). The expansion of CD4+ and CD8+ truncLT-specific T cells, their phenotype, functional capacities concerning cytokine secretion and cytotoxicity, and repetitive expandability will be addressed. From the expanded T cells, a truncLT-specific TCR will be cloned as tool to reprogram T cells for short-term stimulations. After possible combinations and concentration ranges have been narrowed, we will switch from healthy donor blood to that of MCC patients, because the T cells, which were preconditioned by the tumor, may behave differently. In parallel, we will examine the expression of the inhibitory receptors targeted by checkpoint-blockade on truncLT-specific patient-derived T cells.