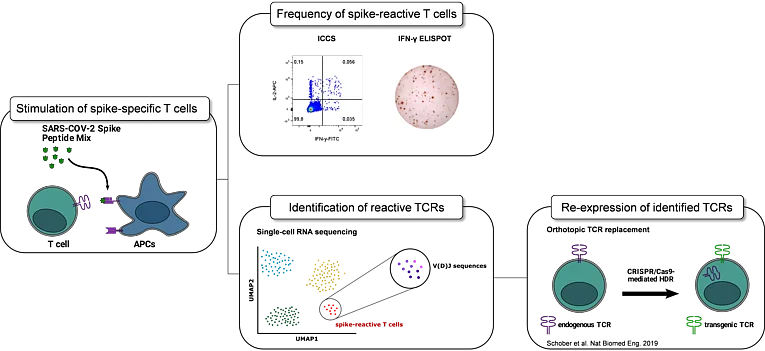
Longitudinal characterization of TCR repertoire functionality after SARS-CoV-2 vaccination
Since the first cases in December 2019, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causing coronavirus disease 2019 (Covid-19) has spread worldwide, infecting millions of individuals. As a response, laboratories and pharmaceutical companies, including BioNTech and Pfizer, developed novel vaccines for protection against severe Covid-19. The mRNA-based Pfizer/BioNTech vaccine BNT162b2 promotes both humoral and cell-mediated antiviral mechanisms by encoding the full-length SARS-CoV-2 spike glycoprotein, which has been shown to elicit antibody as well as T cell responses. Besides its medical importance, the current vaccination effort also provides a rare research opportunity to follow adaptive immune responses of individuals pre- and post-antigen exposure in a systematic manner. This project focuses on the longitudinal characterization of antigen-specific T cell responses in response to vaccination, including T cell receptor (TCR) repertoire evolution, and in-depth characterization of reactive TCRs. Previous studies in mouse models have reported that, from a diverse naïve T cell population comprising a repertoire of TCRs with a spectrum of different avidities, an enrichment of high-avidity TCRs occurs during the course of an immune response. In humans, the contribution of different TCR avidities to an antigen-specific T cell response has barely been studied. With a clinical study following 31 individuals pre- and post-vaccination, we want to clarify which TCR functionalities are selected over time and how the recruitment of antigen-reactive clonotypes is dependent on TCR functional avidity in humans. A deeper understanding of how TCR avidities influence the dynamics of a T cell response may facilitate identification of optimal TCRs for effective therapeutic T cell engineering and help to fine-tune vaccines towards an avidity-enriched T cell response.