Computer-based antiviral drug development: targeting CDKs/vCDKs as crucial determinants of cytomegalovirus and Epstein-Barr virus infections
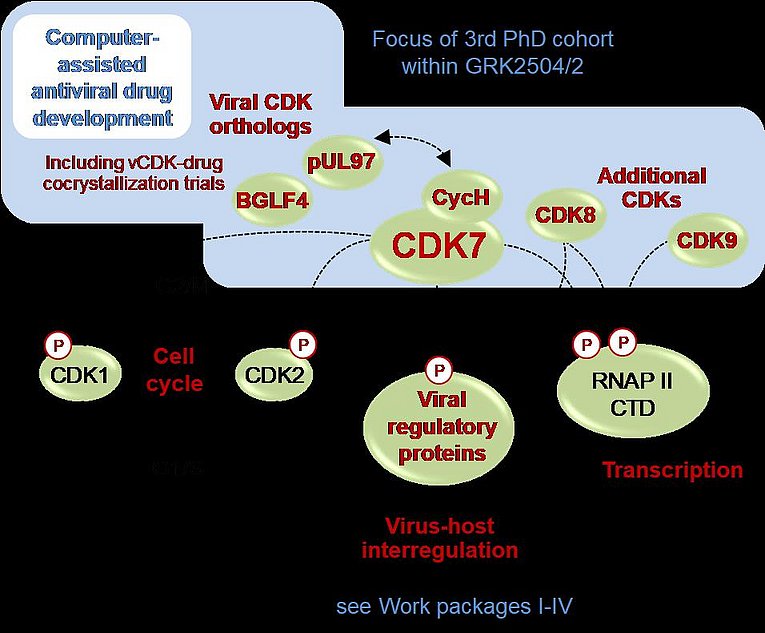
Herpesviruses have evolved effective regulatory properties to modulate the host cell cycle (Schütz et al., 2021). These include reprogramming activities on the normal function of cyclin-dependent kinases (CDKs) towards a virus-supportive role in replication efficiency (Steingruber et al., 2019). The regulatory importance of CDKs for herpesviral replication is crucial for three functional areas (see figure). It was a specific highlight of our studies to see that herpesvirus-encoded protein kinases, such as human cytomegalovirus (HCMV) pUL97 or Epstein-Barr virus (EBV) BGLF4, represent viral CDK orthologs (vCDKs) on the basis of structural and functional similarities (Schütz et al., 2022; Steingruber & Marschall, 2020). Even more striking was the identification of a number of substrate proteins being dually phosphorylated by both CDKs and vCDKs simultaneously, sometimes even at identical substrate phosphosites (Couté et al., 2020). This underlines the strategy of drug targeting towards host CDKs and vCDKs. Dual phosphosubstrates are vCDK/pUL97 itself, pUL69 and pUL50-pUL53 (the core nuclear egress complex presently studied in the 2nd PhD project). Recently, a first pUL97-specific inhibitor (maribavir) has been clinically approved for anti-HCMV therapy. Our work aims to extend the targeting options against HCMV pUL97 and EBV BGLF4 (Wild et al., 2021; Wild et al., 2022; Hutterer et al., 2016) since no anti-EBV treatment is available so far. Our approach is mainly based on computer-assisted methods of antiviral drug development focusing on CDKs/vCDKs. In parallel, crystallization approaches are optimised with recombinant vCDKs to support the structure-based in silico modelling. In essence, such CDK/vCDK-specific small molecules shall be utilised for both, virus-selective or broad-spectrum antiherpesviral drug development.
In order to achieve a goal-oriented development of antiviral kinase inhibitors, we will use approaches of structural bioinformatics and molecular virology. Thereby, we will start with structural models of the vCDKs pUL97 and BGLF4 to identify, on the one hand, small molecules binding to the ATP- and substrate-binding pocket of vCDKs, and to design, on the other hand, inhibitory peptides that bind to the vCDKs competitively at the cyclin interface. This novel antiviral targeting concept will be approached as follows: WP I, Structural modelling of the vCDKs pUL97 and BGLF4 (based on structures of CDKs and other kinases); WP II, Rationalise the binding mode of known kinase inhibitors and predict novel inhibitors of vCDKs by developing a suitable docking protocol; WP III, Structural modelling of the vCDK–cyclin complexes to identify the interaction interfaces and contact-conferring amino acids in these interfaces by molecular dynamics (MD) simulations; WP IV, Using the MD simulation results to predict inhibitory peptides as competitive binders at the vCDK-cyclin interface; WP V, Comparative study to examine the stability of the protein-protein interaction between the vCDKs pUL97 and BGLF4 and the different inhibitory peptides by various MD simulations and the analysis of the force and energy components of the interaction; WP VI, Validate the antiviral properties of this next generation of inhibitors by virological cultured-cell and mouse infectionstudies. Ultimately, the project aims at the computer-assisted prediction and experimental confirmation of small molecules as vCDK-directed anti-EBV and anti-HCMV model drugs.




