Enhancing antiviral immunity by blocking SAMHD1 and targeting viral immune evasion
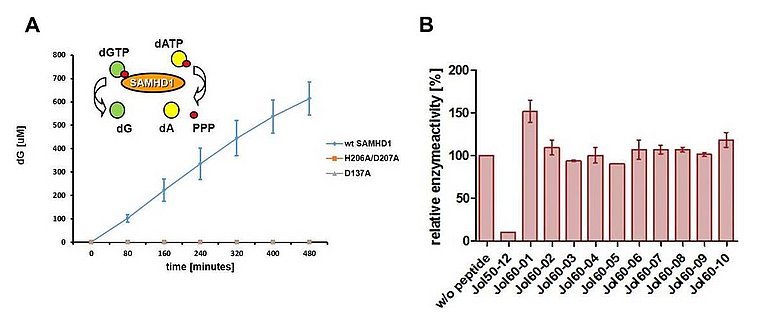
Vpx is essential for efficient SIV infection of Macrophages (Mac) and Dendritic Cells (DC) by mediating the proteasomal degradation of the restriction factor SAMHD1. Functional SAMHD1 forms a tetramer and acts as a dNTP triphosphohydrolase. It cleaves dNTPs and is therefore thought to inhibit retroviral reverse transcription (RT) by reducing intracellular dNTP levels. Preincubation of cells with Vpx-containing virus-like particles (VLP) not only enhances the infection of SIV but also of HIV-1. Since Mac are considered main target cells of HIV-1 in vivo, it is puzzling why HIV-1 does not encode Vpx to counteract SAMHD1 to infect Mac more efficiently. However, recent studies show that the enhancement of HIV-1 infection by Vpx triggers an innate immune response in DCs. This suggests that HIV-1 sacrifices a more efficient Mac infection to evade an innate immune response in DC. Hence, we hypothesized that molecules blocking SAMHD1 activate this immune response to HIV infection. Together with the Eichler lab (Project C1), we generated peptides targeting the tetramer interface of SAMHD1 and already identified a lead peptide that interferes with the dNTPase activity of SAMHD1 in vitro (Fig. 1).
Based on the lead peptide, we will generate modified molecules that block SAMHD1 tetramerisation and determine whether these inhibitors can enhance HIV infection and boost antiviral immunity in DC. The refined peptides will be synthesised and tested in our established in vitro dNTPase assay (Fig. 1). Also, small molecules libraries will be tested in this assay to identify inhibitor candidates. Active candidates will be analysed in HIV infectivity assays on myeloid cell lines and primary cells for their ability to revoke restriction. Next, we will analyse their effect on the innate immune response upon infection by qPCR, FACS, and ELISA. Furthermore, we will take advantage of our previously established SAMHD1 KO mouse model and determine the impact of blocking SAMHD1 in dendritic cells on the induction of an antiviral immune response in vivo. Together, the proposed project will identify inhibitors of the restriction factor SAMHD1 and evaluate the potential of the inhibitors to elicit an enhanced antiviral immune response. This strategy will prove important for the development of adjuvants for HIV vaccines and other lentiviral vector-based vaccine efforts.