Optimizing intrastructural help for virus-like particle vaccine regimens
By the end of 2019, about 38 million people were living with HIV, but only 67% of these patients were receiving antiretroviral therapy. This percentage is an indication of how difficult it is to get access to appropriate therapy. Since there is no cure for HIV infection yet, vaccination against HIV is a promising option.
Previous researches have shown that antibodies in HIV vaccine trials (Vax003, Vax004, RV144) are not as efficient and durable to provide sufficient protection against HIV infection for a lifetime. Beside Env epitope binding, different anti-Env IgG antibody subtypes and their Fc effector functions may also play a role for protectivity.
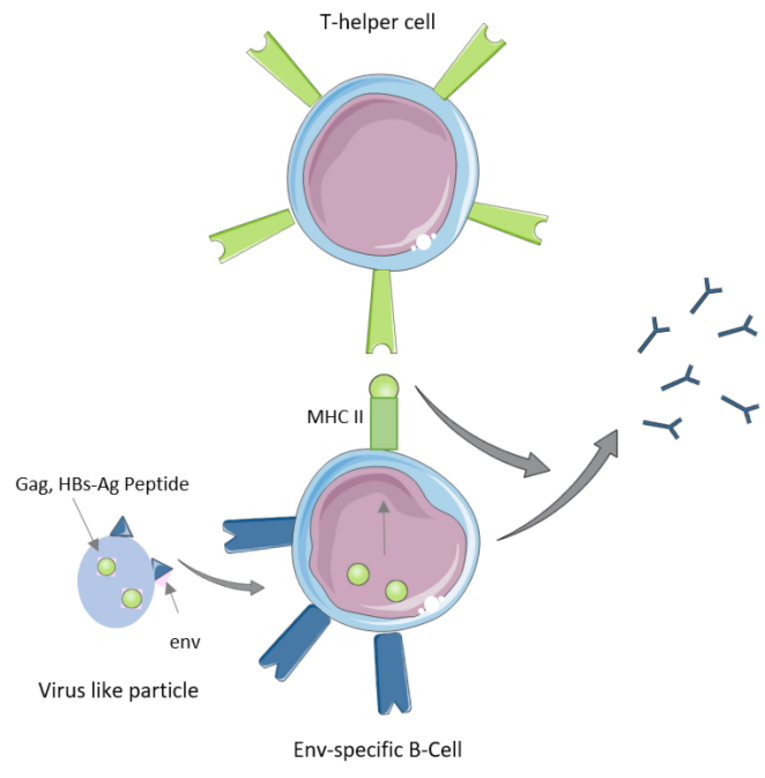
Env vaccination in mice predominantly induces a Th2 response. In the past, the principle of intrastructural help (ISH) was used to increase the Th1 response against Env, which comes with stronger antiviral Fc effector functions by interaction between env-specific B-cells and heterologous T-helper cells. The mechanism of ISH is shown in Figure 1.
In this project, ISH is induced by a prime immunization with Hepatits B surface antigen (HB-sAg) DNA and an HIV virus-like particle (VLP) vaccination. The VLPs contain both env antigens (the envelope glycoprotein gp140 on the surface) and internal structural proteins (gag) fused to T-helper cell epitopes of HB-sAg, allowing Env-specific B cells to be stimulated by T-helper cells by presenting the gag and HB-sAg peptides via MHC II molecules to HB-sAg-specific T-helper cells.
This project focuses on two factors to optimize and better understand env-associated ISH.
I, The optimal time point between prime immunization and VLP boost.
Mice will receive the HB-sAg-DNA prime and HB-sAg epitope-bearing VLP boost with different time intervals in-between (0 d – 28 d). Blood samples will be taken two weeks after the VLP immunizations and the anti-Env IgG subtype distribution will be evaluated by serum ELISAs. This aims at defining the best time point for the VLP boost to achieve a maximum of ISH effects.
II, The influence of different amounts of T-helper cell epitopes per VLP on the ISH phenotype.
We intend to titrate the quantitative amount of HB-sAg epitopes within the VLPs by using different ratios of HB-sAg-Gag-DNA and wildtype Gag-DNA for transfection of the producer cells. The successful HB-sAg titration will be verified by VLP Western Blots. Finally, ISH immunizations will be performed in mice using VLPs with serial dilutions of encapsulated HB-sAg epitopes. This will give insight, whether ISH phenotypes gradually diminish with less epitope availability or whether a certain cutoff for successful induction of ISH can be determined.
Taken together, this project will strengthen the knowledge about the in vivo requirements of ISH and will help to optimize and fine-tune future vaccine applications in the context of ISH.