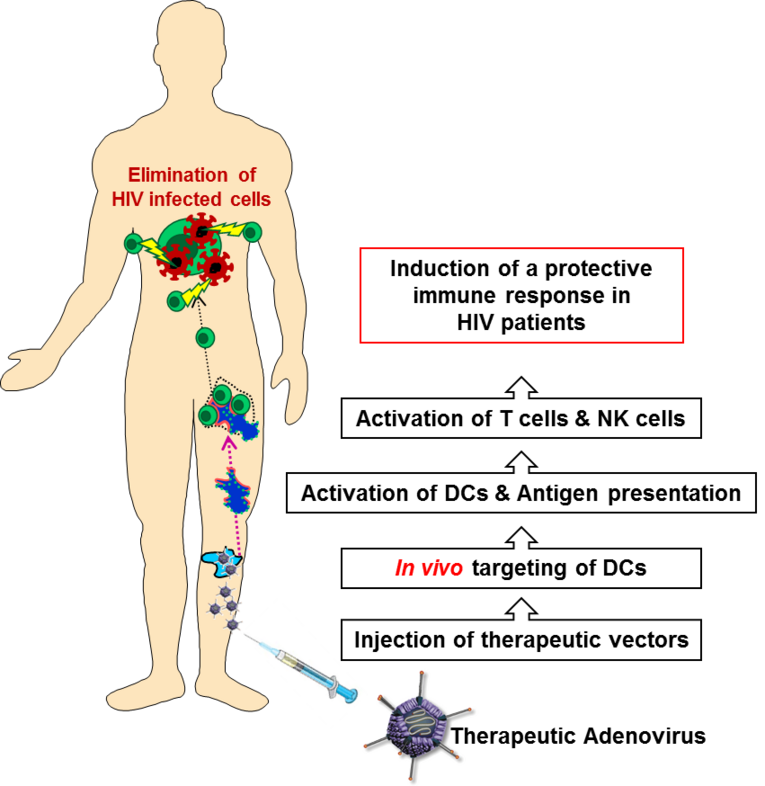
Transcriptional targeting of dendritic cells as a new therapeutic vaccine against HIV-1
Up to date, there is no cure for the human immunodeficiency virus (HIV) available. Treatment with antiretroviral therapy (ART) is expensive and relies on strict adherence of patients. More recent dendritic-cell (DC) based vaccination attempts are superior to classical vaccination strategies regarding their potential to activate the adaptive immune system and presentation of a broad set of antigens. Since ex vivo transduction of DCs appears challenging considering the limited scope for mass production, in vivo targeting strategies appear to be a more suitable approach. This project aims for a new strategy for developing a therapeutic vaccine against HIV based on transcriptional targeting of DCs. Specificity of transcriptional targeting will be achieved by utilizing the human CD83 promoter P-510, which is specifically active in mature but not immature DCs. In short, expression of the gag protein and the immune stimulatory constitutive active (ca)IKK in DCs is expected to increase presentation of gag epitopes to T cells and ultimately result in a protective immune response in the patient. To test this hypothesis, we will follow up on three distinct specific aims:
Aim 1: Generation of therapeutic adenoviral vectors expressing the HIV gag protein and caIKK controlled by the CD83 promoter.
Aim 2: Functional analysis of therapeutic vectors in vitro.
Aim 3: Functional analysis of therapeutic vectors in vivo in a humanised mouse model.
With this project, we hope to contribute to increased understanding of the immune response to HIV infection and, in the long term, add valuable input to vaccination attempts in HIV patients.