Figure was created with BioRender.
Longitudinal analysis of of HIV-specific TCR clonotype expression, avidity and function in peripheral blood and lymph nodes
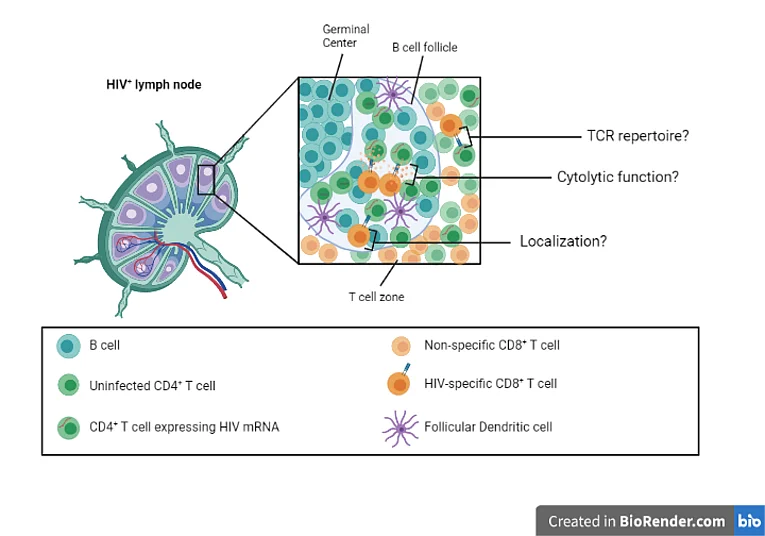
Currently, there are close to 38 million people living with HIV infection worldwide and no functional cure is available at present. Combination antiretroviral treatment (ART) has drastically improved life expectancy and quality of life for people living with HIV, but despite its effectiveness in suppressing plasma viral loads latently infected cells persist in various tissue sites. The majority of cells harbouring integrated provirus are found in lymphoid tissues, which have been difficult to study. Achieving a functional cure for HIV will require induction of immune cell responses that are able to target infected cells expressing viral proteins within the lymphoid tissue compartment. However, the nature of the antiviral immune responses within lymphoid tissues remains to be fully defined. Particularly important information may derive from the rare subset of individuals that naturally control HIV infection without the need for ART. Based on preliminary data generated in the lab, we hypothesize that highly functional cytotoxic T lymphocyte (CTL) responses are critical for durable immune control of HIV, and are actually able to reduce and potentially eliminate the replication competent reservoir. Here, we propose to define the functionality of CTL within the lymph node microenvironment through examination of excisional lymph node biopsies from persons who spontaneously control viremia compared to progressors, and also examine the impact of immediate therapy of hyperacute HIV infection on these responses. We will elaborate on recent findings suggesting that CTLs in lymph nodes of people that naturally control HIV infection achieve control via non-cytolytic mechanisms by examining both cytolytic and non-cytolytic functions as well as performing transcriptional analysis on lymphoid and peripheral blood CTLs. We will further examine the T cell receptor (TCR) repertoire and identify tissue-specific changes in the presence and frequency of T cell clones. Finally, we will use in situ hybridization and in situ sequencing to analyze the spatial localization of HIV-specific T cells within lymph node tissue sections by microscopy. The detailed characterization of the CTL function, the clonotype repertoire and their localization within the lymph node will be correlated to the size of the replication-competent reservoir in this compartment to identify responses that may be beneficial to induce with immunotherapy.