An animal model of HLA-independent, chimeric antigen receptor-mediated adoptive immunotherapy against cytomegaloviruses
Allogeneic stem cell transplantation (SCT) is currently the therapy of choice for children with high-risk leukaemia in first remission and patients with recurrent disease. Viral infection, in particular with cytomegalovirus (CMV), or reactivation of latent CMV infection continues to be a frequent and life-threatening complication of SCT. An efficient T-cell immune response is imperative in long-term control of CMV infection. Currently, adoptive T‑cell therapies using effector cells retargeted by chimeric antigen receptors (CAR) toward CD19+ tumour cells show very promising results in the treatment of leukaemia.
The projects builds on our previous description of the first functional CMV-specific CAR, composed of a CMV glycoprotein B (gB)-specific single chain variable domain (scFv) linked to the signalling domains of human CD3z and co-stimulatory domains of CD28. Transient expression using electroporation of CAR-encoding mRNA induced efficient lysis of CMV gB-expressing cells, degranulation and secretion of effector cytokines (Full et al., 2010; Lehner et al., 2012). In addition, we recently uncovered an intrinsic resistance of CMV-infected cells to cytotoxic T lymphocyte (CTL)-mediated killing, affecting both HLA-independent (CAR) and dependent (TCR) recognition pathways (Proff et al., 2016). Nevertheless, we have evidence that this does not preclude in vitro antiviral activity mediated by cytokines such as TNF and IFNg secreted by CAR-CTL (Proff et al., 2018).
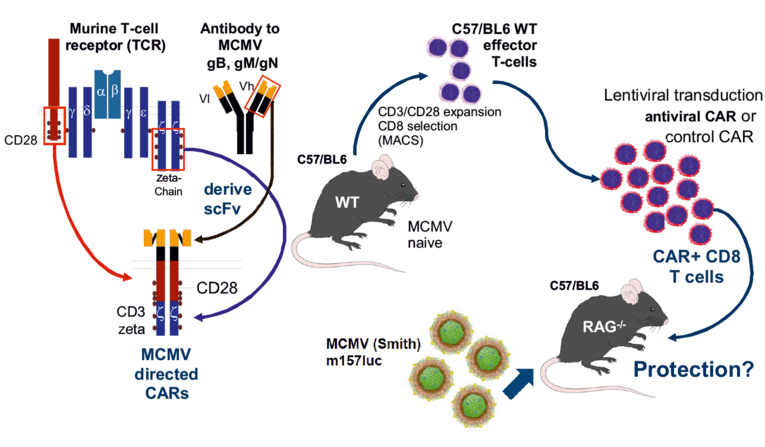
Here we will test whether CAR-retargeted T-cells can restrict viral infection in the murine CMV (MCMV) infection model. The proposed project will use the immune deficient RAG-/- mice and MCMV engineered to express a luciferase reporter, MCMVm157luc (established by Thomas Winkler, C3). MCMV-specific murine CARs will be designed from antibodies directed against MCMV glycoproteins expressed on the surface on infected cells. scFv derived from sequences of murine monoclonal antibodies against MCMV gB and gM/gN (will be combined into murinised versions of antiviral CARs (Figure, left). These CARs will then be transduced into murine cytotoxic effector T-cells by lentiviral vectors. Redirected cells will be functionally validated in vitro (target lysis, degranulation, cytokine secretion), and the most effective CARs will then be analysed in the mouse model (Figure, right; viral spread by bioluminescence, viral organ load). In addition, our published human CMV gB-targeting CAR will be tested in vivo in RAG-/- mice infected with a MCMVm157luc variant ectopically expressing HCMV gB. The project’s goal is to validate (or refute) the hypothesis that a CAR-mediated adoptive antiviral immunotherapy can restrict viral infection in the relevant MCMV animal model.
Provided this project proves the efficiency of an antiviral CAR against MCMV, further investigations can test whether additional CAR against further MCMV target molecules (gH, gL), or other costimulatory signalling components of CAR, could enhance the efficacy of this approach. Finally, this approach can be translated into an HLA-independent patient-specific immunotherapy of human CMV infection by employing T-cells transfected with readily available, “off-the-shelf“ CAR mRNA. This approach may reduce viral complications of allogeneic SCT, particularly in CMV-mismatched, high-risk allogeneic SCT (CMV-seronegative donor/seropositive recipient).