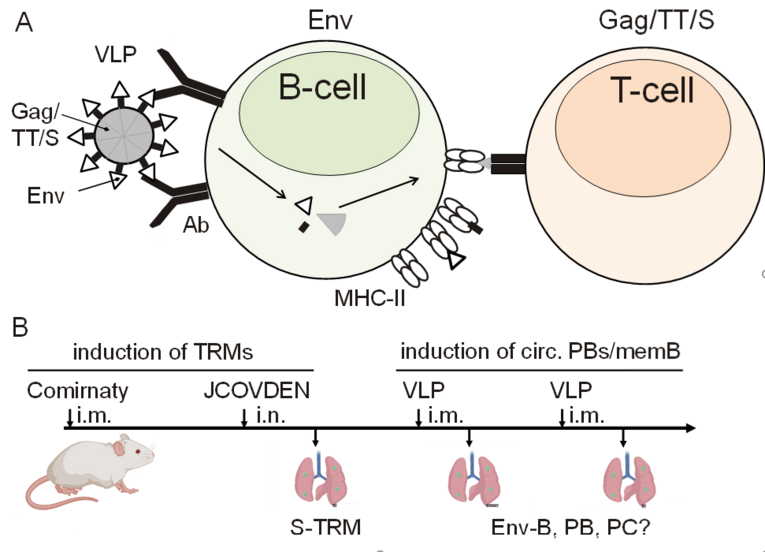
Improving mucosal immunity by intrastructural help
The best possible protection induced by vaccination blocks or controls viral replication at the portal of viral entry. Sterilising immunity can be achieved by passive immunisation with neutralising antibodies. However, inducing long-lasting protective levels of neutralising antibodies by active immunisation remains a challenge at least for HIV as well as a number of respiratory viruses. Induction of mucosal immune responses may be key for fast and efficient control of viral replication at the mucosal entry site. We have previously shown that a combination of systemic priming immunisations followed by mucosal booster immunisations efficiently induces tissue-resident CD4+ and CD8+ T cell responses in the respiratory tract and provides protection from viral challenge (Lapuente et al., 2021). Induction of tissue-resident B cell (TRB) responses by vaccination and their relevance for protection has been rarely studied (reviewed in Lee et al., 2022). We have previously developed a systemic immunisation regimen with HIV virus-like particle (VLP) vaccines, which allow modulating HIV Env B cell responses by “preexisting” T helper cells against internal antigens of the VLPs (Nabi et al., 2013; Storcksdieck et al., 2015). Since naive Env-specific B cells can internalise and process entire VLPs in a BCR and Env dependent manner, T helper cells specific for internal viral proteins (e.g. Gag) can provide help for Env-specific B cells (see figure A). Priming mice with Tetanol prior to booster immunisations with VLPs incorporating a T-helper cell epitope of tetanus toxoid (TT) also increased the Env antibody response (Elsayed et al., 2018). This indicates that T helper cells induced by licensed vaccines can be harnessed to optimise the Env antibody response to particulate vaccines. Bioinformatics analyses will allow dissecting this response at the single-cell (sc) level (Culemann et al., 2019).
Since mucosal vaccine delivery meets a number of practical hurdles, the project aims to explore intrastructural help by “preexisting” tissue-resident T helper cells (hTRM) as a general strategy to recruit circulating memory B cells or plasmablasts (PB) to the mucosa of the respiratory tract. hTRMs in lungs will be induced by an intramuscular Comirnaty prime – nasal Jcovden boost immunisation regimen (see figure B). Mice will then be immunised intramuscularly with HIV-VLPs containing HIV Env on their surface and an immunodominant T helper cell epitope from the spike protein (S) of SARS-Cov-2 inside. The HIV Env protein is used in these experiments as a model antigen due to the availability of reagents and established methods. Four to six days after a first intramuscular VLP immunisation, Env-specific acute PBs presenting the S T helper cell epitope are expected to circulate systemically. This allows analysing whether the S-specific hTRMs direct the circulating PBs for residency in the lung tissue. Similarly, the trapping of Env specific memory B cells in the lungs can be explored after a second intramuscular VLP immunisation. Mice immunised with VLPs lacking the S T helper cell epitope will serve as controls to nail down the relevance of cognate T cell help. As a benchmark, induction of Env-specific TRB responses will also be studied after systemic prime nasal boost immunisations with Env encoding vaccines. TRB responses in the lung will be analysed by flow cytometric sorting of tissue-derived B, PB and plasma cells (PC) negative for a vascular staining marker and multiomics single cell analyses. Transfer of marked BCR-transgenic B cells specific for Env and identification of Env-specific mucosal B cells, PBs and PCs by virtue of their BCR sequences identified in the same animal from systemic Env specific memory B cells will allow focusing the analysis on the Env specific TRB response.