Induction of tissue-resident memory T cells by gene-based vaccines as first line of defence against pathogens entering the host via mucosal surfaces
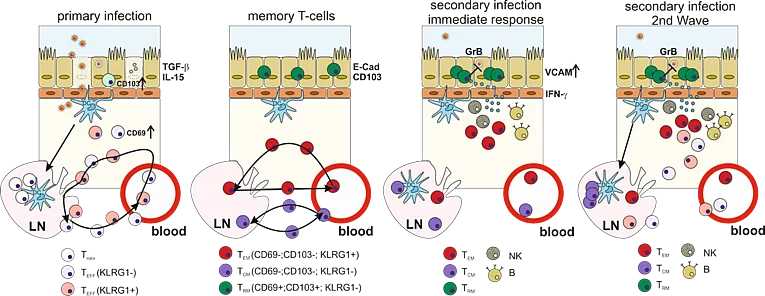
The protective capacity of a vaccine critically depends not only on the form of the antigen/pathogen (inactivated or live-attenuated) but also on the location where it enters the host. Accordingly, an effective vaccine against respiratory tract infection should mount a strong mucosal immunity including mucosal IgA antibody responses and the induction of tissue-resident memory cells (TRM) in the lungs. The TRM represent a type of memory cells which do not circulate like central memory (TCM) or effector memory T-cells (TEM), but remain in non-lymphatic tissues, like the skin, brain or the lungs. These front line immune cells provide immediate effector functions upon secondary pathogen encounter have a distinct role in recruiting circulating T-cells and even induce an innate immunity-like antiviral state. In mice, CD69+CD103+ TRM induced by a primary intranasal Influenza A Virus (IAV) infection reside in the lung and provide heterosubtypic immunity against secondary infections with divergent IAV strains, which cannot be observed after vaccination with inactivated vaccines.
In the past years, our group worked intensively on the optimisation of gene-based vaccines against viral infections (HIV, IAV, RSV). We established protocols for the mucosal and systemic delivery of DNA and adenoviral vector vaccines resulting in efficient protection against challenge infections with IAV or RSV in mice and non-human primates (Grunwald et al., 2014; Lapuente et al., 2018a; 2018b).
The goal of this project is the development of gene-based vaccines able to induce protective, long-lasting TRM responses at a mucosal barrier tissue like the lung. We utilise gene-based vaccines to simulate a natural infection by expressing not only the viral antigens, but also innate immune signalling molecules induced by IAV. Tissue-specific gene-expression profiles, cytokine/chemokine levels as well as migrating immune cells including different APC populations will be analysed in depth early after vaccination and correlated to TRM formation in the lungs and vaccine efficacy. Characterisation of the underlying immunological mechanisms by appropriate knock-out models will guide further vaccine optimisation, e.g. by harnessing DC-targeting strategies or co-expression of essential signalling molecules.