Characterization of viral reservoirs in HIV-infected children undergoing ART and bNAb treatment
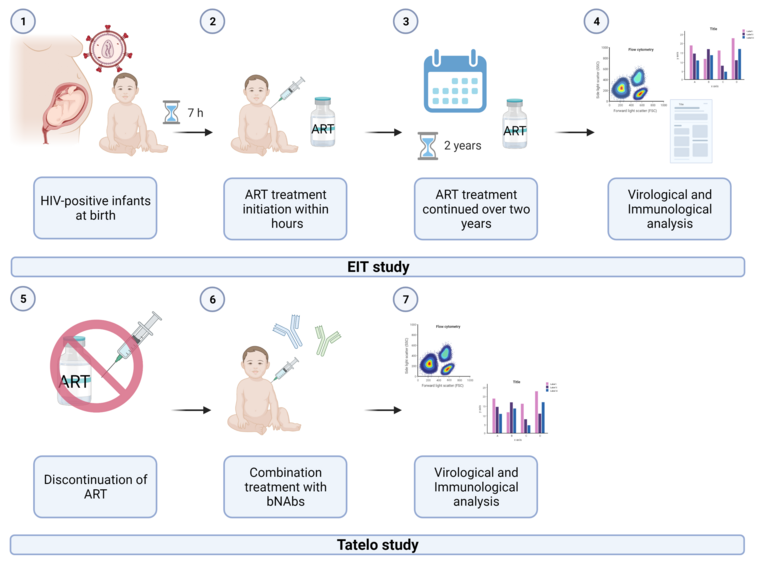
Despite the effective reduction of mother-to-child transmission of the human immunodeficiency virus (HIV), neonatal HIV-1 infection remains a public health problem, with 160.000 infants newly infected in 2018. Clinical studies suggest a more rapid disease progression with severe outcomes in HIV-infected infants compared to adults when left untreated. Due to that, the WHO recommends early anti-retroviral therapy (ART) treatment initiation during the first weeks of life. Our EIT study showed that very early ART initiation within the first hours of life can limit the viral reservoir formation and prevent irreversible damage to the neonatal immune system. However, ART is difficult to maintain over a lifetime, and ART's long-term toxicities are only beginning to be understood. Furthermore, resistance mutations can lead to therapy failure and thereby cause viral rebound. This creates a need for novel strategies to maintain effective viral control during periods of ART discontinuation. A proposed strategy is immunotherapy with broadly neutralizing antibodies (bNAbs), which effectively reduced plasma viremia and delayed viral rebound upon ART treatment interruptions in adults. This project focuses on a clinical study, subjecting the cohort of very early treated children (EIT cohort) to a combination treatment of two bNAbs while ART is discontinued (Figure 1). We want to gain insight into the impact of this immune therapy on the clinical outcome, size and distribution of the viral reservoir, and immune responses of the neonatal immune system.