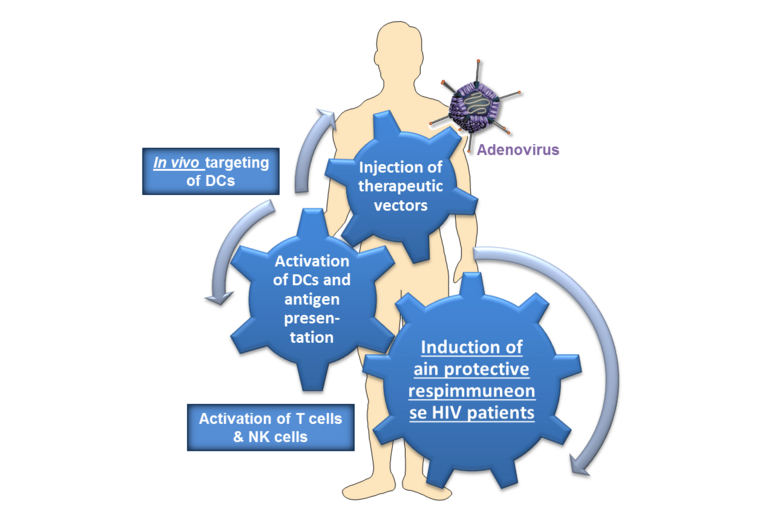
Transcriptional targeting of dendritic cells as a new therapeutic vaccine against HIV-1
Today, over 38 million people are infected with HIV worldwide, 1.2 million of them dying each year. Although combined antiretroviral therapy (cART) is highly effective to prevent clinical progression and death, it is unable to eradicate the infection, thus necessitating therapy throughout life. Moreover, viral resistance and adverse effects in the medium-to-long term as well as costs are important limitations for lifelong adherence to this therapy (Garcia et al., 2013). Therefore, for an effective control of the epidemic, new cost-effective and viable therapeutic strategies need to be evaluated. Towards this goal, a cell-based vaccine using in vivo targeting of DCs represents a highly attractive strategy. Thus, the long-term aim of this project is the development of a new cost-effective therapeutic vaccination therapy for HIV-1 infected individuals by the direct transcriptional in vivo targeting of mature DCs in patients (Fig. 1). The human CD83 promoter complex has been previously identified and characterized at the molecular level in our laboratory (Stein et al., 2013). CD83 is not expressed by immature, tolerogenic DCs, but is highly induced in mature, immunogenic DCs. Hence, the CD83 promoter complex represents an ideal candidate for transcriptional targeting of mature DCs in vivo (Knippertz et al., 2016). Therefore, under the control of the CD83 promoter, we will express virus specific, immunogenic HIV antigens in combination with immune stimulatory molecules.
For the proof-of-concept we will simultaneously express a viral model antigen as well as immune stimulatory molecules, or a co-stimulatory molecule under the control of the CD83 promoter complex in order to further boost the antiviral immune response. Additionally, viral antigens will be tagged for the ubiquitin proteasome system to improve presentation via MHC class I. Furthermore, we will combine transcriptional targeting with transductional targeting via a specific molecule on the cell surface of DCs. Next, we will perform in vitro functional assays of Ad-transduced DCs regarding their migratory capacity, cytokine secretion as well as their ability to stimulate CD4+ T cells or to prime naïve CD8+ T cells in an antigen-specific manner to prove the concept. Subsequently, we will perform in vivo experiments in order to analyse the activity of the therapeutic Ads using human hemato-lymphoid-system (HHLS) mice, which will allow for the characterisation of cells with specific CD83 promoter activity in vivo. Finally, this new strategy aims to target mature DCs in vivo and in the long term to induce anti-viral immune responses directly in patients, thereby circumventing the currently used, laborious and cost intensive ex vivo generation and manipulation of DCs.