Structure-based design and optimisation of ligands for novel antiviral strategies
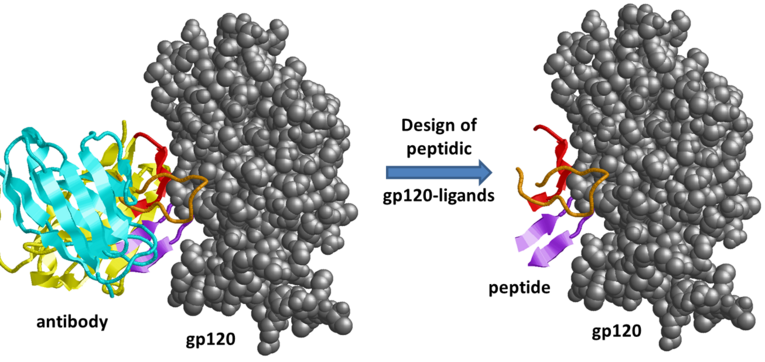
Broadly neutralising antibodies targeting the fusion proteins gp41/gp120 represent a promising strategy for protection from HIV-1 infection. Such antibodies can be used for passive immunisation and are currently tested in clinical trials, but they are expensive and difficult to produce. As an alternative, the antibody-antigen recognition can be mimicked by synthetic peptides, which has already been demonstrated to be a successful concept for a range of diverse protein interactions. The project aims to transfer the favourable binding properties of gp120-neutralising antibodies to small peptide ligands derived from the antigen-binding site of the respective antibodies.
For that purpose, a comprehensive structural analysis of gp41/gp120-antibody complexes will be performed. This analysis will include the length and spatial arrangement of the complementarity determining regions (CDRs) as well as the identification of key residues for antigen binding. These analyses of static protein structures will be supplemented by molecular dynamics simulations, which give additional insight into the conformational stability of the complexes and the role of glycan moieties for the recognition process.
The most promising epitopes will be modelled as peptides and their binding properties and conformational stability of these designed ligands will be further investigated by molecular dynamics simulations. The synthesis and experimental verification of the binding affinity of these ligands will be performed in the group of Prof. Jutta Eichler. Ligands that exhibit favourable properties will be further computationally optimised regarding their binding affinity and specificity. In this step, we will optimise the sequence to stabilise peptide conformation and to maximise intermolecular contacts.
To further enhance the functional properties, we plan to design hybrid ligands by combining high-affinity CDRs from different antibodies. A particular focus will be on those CDRs that contact spatially proximal regions of trimeric pre-fusion HIV-1 Env. This work will also include optimisation of the length and flexibility of the connecting linkers. The structural properties of all constructs will be further verified by molecular dynamics simulations and then be subjected to experimental testing by the experimental groups of this research collaborative. Taken together, we expect that the strategy outlined above will allow the design of small potent ligands that are capable to interfere with the virus-host interaction.



