CMV-specific T cells for prevention and therapy of CMV reactivation in patients after kidney transplantation
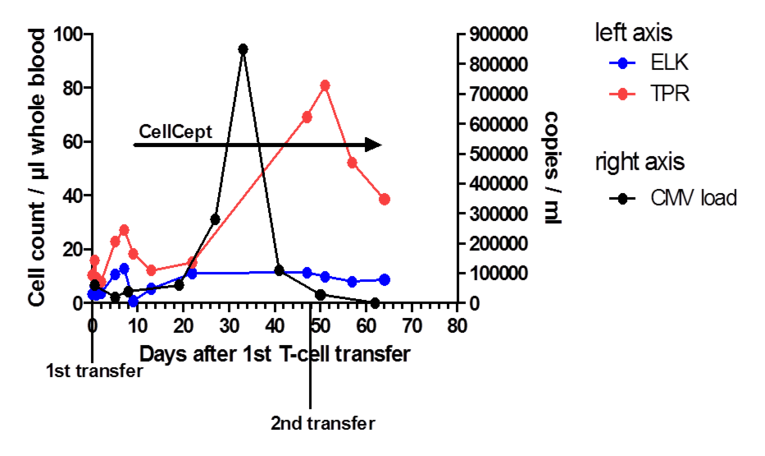
Cytomegalovirus (CMV) reactivation and infection still represents a major problem after kidney transplantation. Despite the possibility of effective prophylaxis, primary infection or viral reactivation often occurs after discontinuing medication. On the one hand, immunosuppression is regularly compromised because of CMV infection, also leading to an increased risk of acute graft rejection. On the other hand, T-cell maturation and activation is impaired because of the treatment with T-cell depleting antibodies and calcineurin or mTOR inhibitors to reduce the risk of graft rejection and thereby negatively affecting the surveillance of CMV. Moreover, prophylaxis, surveillance and treatment of CMV (Reischig, 2012; Fishman et al., 2007) are expensive and associated with severe side effects. A GMP-compliant protocol for the generation of CMV- and EBV-specific peptide-stimulated T-cells for patients after allogeneic stem cell transplantation (aSCT) has been established by our GMP group (UKER, Medizin 5; Gary et al., 2018). The protocol is used for the stimulation of cells from CMV- and EBV-seropositive, HLA-matched allogeneic donors covering defined virus epitopes. In 2014, a phase I/IIa clinical trial based on this protocol was approved by the federal authorities for the preemptive and preventive adoptive transfer of CMV/EBV-specific cells after aSCT. The use of CMV-specific cells after kidney transplantation has been reported in a case study (Macesic et al., 2015). As individualized treatment, CMV-specific T-cells were generated by our GMP group from the apheresis product of an immunosuppressed kidney-transplanted patient suffering from ganciclovir-resistant CMV reactivation and transferred. Subsequently, virus-specific T-cells could be detected in the in the patient’s peripheral blood (Fig. 1) coinciding with CMV suppression, providing a first hint for the feasibility of the project.
T-cell therapy may be a powerful tool against CMV infection and reactivation after kidney transplantation. Therefore, establishing how CMV-specific cells from patients with end-stage renal failure can be optimally expanded is a central point of the project. Most importantly, the protocol will be modified to serve CMV-negative patients. Furthermore, this project will focus on the characterization of potential alloreactivity of the T-cell products against the allograft. The long-term goal is the preparation of a clinical protocol for prophylactic/pre-emptive adoptive CMV-specific T-cell therapy in patients after kidney transplantation.



