Antiretroviral therapy with amendment of HIV-1-specific T-cell response
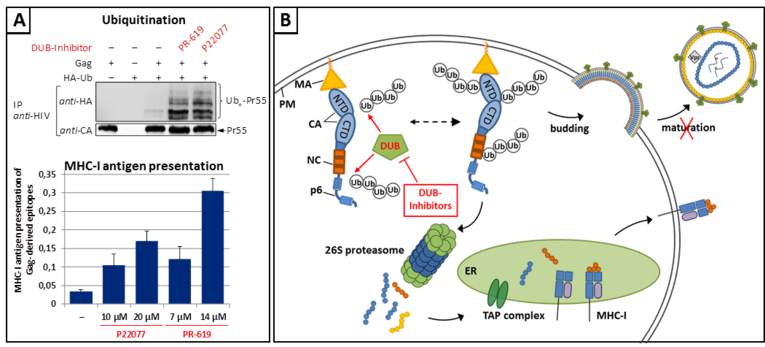
Continuous optimization of both, antiretroviral therapy (ART) and efficient prophylactic vaccination strategy, are crucial to ultimately combat the HIV pandemic. Induction of a potent and specific CD8+ T-cell response against epitopes derived from the major structural HIV-1 protein Gag is an important part of this ongoing endeavour. Our longstanding research on this topic reveals that three major components of the ubiquitin proteasome system (UPS) - the proteasome holoenzyme, ubiquitin ligases, and deubiquitinating enzymes (DUBs) - play a crucial role in virus replication as well as in the regulation of the immunogenicity of Gag. We and others have been elaborating the working hypothesis that the metabolic stability of an antigen inversely correlates with its entry into the UPS and the MHC-I pathway, and thus its ability to induce an efficient and specific CD8+ T-cell response. This assumption is based on the general dogma that the majority of the MHC-I epitopes is derived from so-called defective ribosomal products (DRiPs). DRiPs are de novo synthesized erroneous proteins which are rapidly degraded via the UPS. Originally, we showed that Gag represents a bona fide substrate for the DRiP pathway, and that certain motifs within the C-terminal p6 protein of HIV-1 Gag regulate its entry into this pathway. These observations led us to hypothesize that specific DUBs bind to p6 and regulate the ubiquitination status of Gag and thus its entry into the DRiP- and MHC-I pathway. Indeed, we confirmed that certain small drug DUB inhibitors can both (i) inhibit HIV-1 replication and (ii) enhance the ubiquitination, and thus the MHC-I presentation of Gag-derived epitopes.
The aim of the project is to elucidate the mechanism how DUB inhibitors stimulate the entry of Gag into the MHC-I pathway, and thus augment the immune recognition of HIV-1-infected cells by CD8+ T-cells. First, we want to investigate the influence of DUB inhibitors on the entry of Gag into the DRiP pathway. Second, we will assess whether DUB inhibitors have any positive influence on the proliferation of immune cells. Finally, we will seek proof-of-concept that DUB inhibitors enhance immune recognition of HIV+ cells by CD8+ T-cells. T-cell receptor (TCR) transgenic T-cells will be stimulated in vitro and in vivo in the presence of DUB inhibitors. To explore a potential application of DUB inhibitors as T-cell adjuvants, their influence on CD8+ T-cell responses induced by vaccination will be studied in OT-1 or Balb/c mouse models.